|
Ĭarbon's crystal structure is hexagonal at high pressures and temperatures it forms diamond (see below). Silicon melts at 1414 ☌, germanium melts at 939 ☌, tin melts at 232 ☌, and lead melts at 328 ☌. The melting points of the carbon group elements have roughly the same trend as their boiling points. Carbon, the lightest carbon group element, sublimes at 3825 ☌. The boiling points of the carbon group tend to get lower with the heavier elements. There are no known compounds of flerovium. Lead forms four oxides, a sulfide, a selenide, and a telluride. Lead forms dihalides and tetrahalides with fluorine and chlorine, and forms a dibromide and diiodide, although the tetrabromide and tetraiodide of lead are unstable. Lead forms one hydride, which has the formula PbH 4. Tin forms chalcogenides with one of each naturally occurring chalcogen except polonium, and forms chalcogenides with two of each naturally occurring chalcogen except polonium and tellurium. Tin forms dihalides and tetrahalides with all halogens except astatine. Tin forms two hydrides: SnH 4 and Sn 2H 6. Germanium nitride has the formula Ge 3N 4. Germanium bonds to all natural single chalcogens except polonium, and forms dioxides, disulfides, and diselenides. Germanium forms tetrahalides with all halogens except astatine and forms dihalides with all halogens except bromine and astatine. The first two germanium hydrides are GeH 4 and Ge 2H 6. Silicon also forms a dioxide and a disulfide. Silicon forms tetrahalides with fluorine, chlorine, bromine, and iodine. Silicon forms several hydrides two of them are SiH 4 and Si 2H 6. Carbon also forms many oxides such as carbon monoxide, carbon suboxide, and carbon dioxide. Although tin is chemically a metal, its α allotrope looks more like germanium than like a metal and it is a poor electric conductor.Ĭarbon forms tetrahalides with all the halogens. Tin and lead are both capable of forming +2 ions. Tin and lead both are metals, while flerovium is a synthetic, radioactive (its half life is very short, only 1.9 seconds) element that may have a few noble gas-like properties, though it is still most likely a post-transition metal. Silicon and germanium, both metalloids, each can form +4 ions. The tendency to lose electrons increases as the size of the atom increases, as it does with increasing atomic number.Ĭarbon alone forms negative ions, in the form of carbide (C 4−) ions. Double bonds are characteristic for carbon ( alkenes, CO 2.) the same for π-systems in general. For single bonds, a typical arrangement has four pairs of sp 3 electrons, although other cases exist too, such as three sp 2 pairs in graphene and graphite. Bonds in these elements often lead to hybridisation where distinct s and p characters of the orbitals are erased. 
These elements, especially carbon and silicon, have a strong propensity for covalent bonding, which usually brings the outer shell to eight electrons. An isolated, neutral group 14 atom has the s 2 p 2 configuration in the ground state. 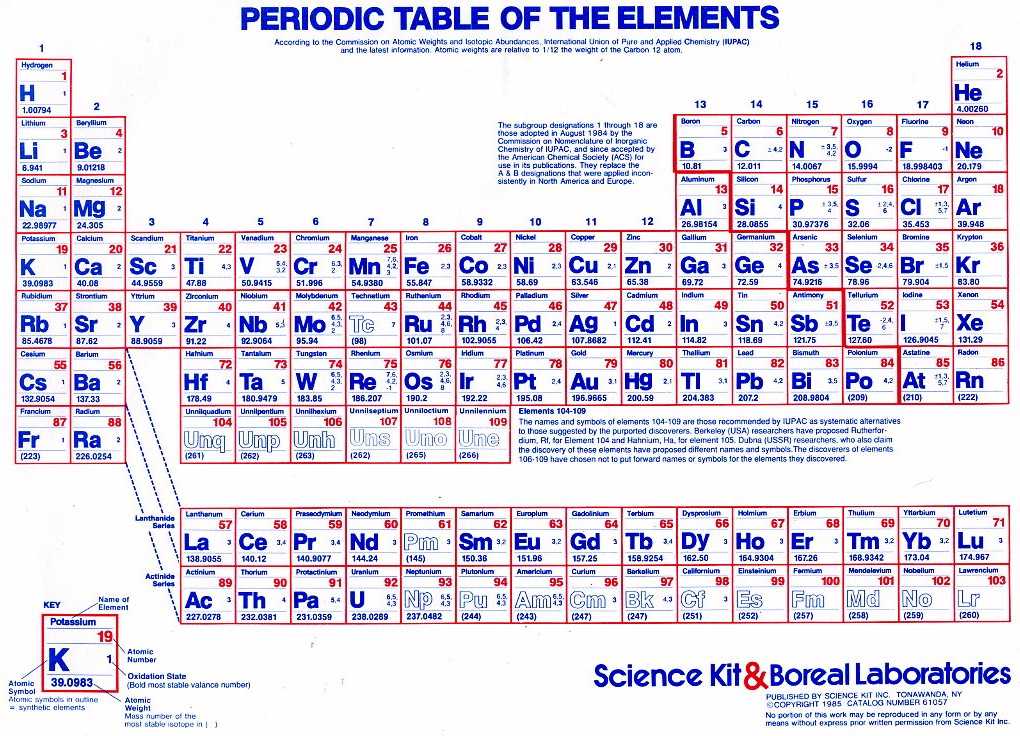
Like other groups, the members of this family show patterns in electron configuration, especially in the outermost shells, resulting in trends in chemical behavior:Įach of the elements in this group has 4 electrons in its outer shell. They are also known as the crystallogens or adamantogens. The group is also known as the tetrels (from the Greek word tetra, which means four), stemming from the Roman numeral IV in the group names, or (not coincidentally) from the fact that these elements have four valence electrons (see below). In the field of semiconductor physics, it is still universally called group IV. In modern IUPAC notation, it is called group 14. The carbon group is a periodic table group consisting of carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and flerovium (Fl).
0 Comments
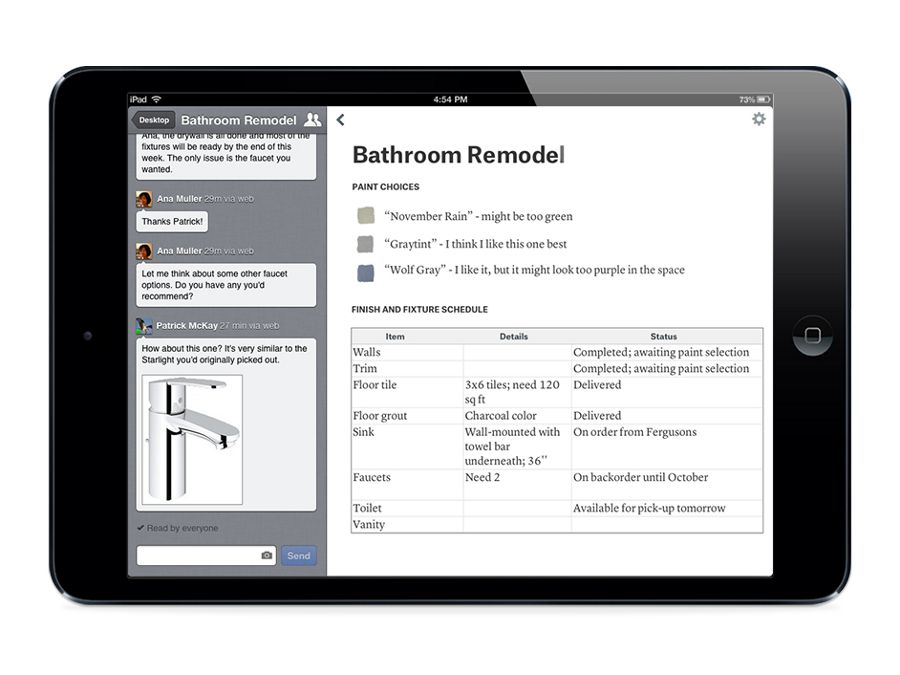
Using quip makes the process of writing and sharing documentation a lot more painless and enjoyable. Overall my experience has been very positive. It's a bit of an irritant to correct that every time. Also, have noticed when I copy paste code into code-blocks, the pasted content does not fit within the default page-width, but creates a horizontal scroll-bar within the code-block. Although it has the ability to add spreadsheets, I don't use it much. Quip requires the internet and often I forget that I have to be connected to save changes. It also has the ability to create private folders which I use to store work in progress ideas and the ability to restrict access to some files / folders to specific people. It also prevents unnecessary emails as users can chat / comment directly on quip. The ability to have one central location from where all of us can access it makes this a huge time saver for all. Product feature requirements are dynamic - in that, they change often. I use quip mainly to organize my work and collaborate with my team members in documenting feature requirements. Productivity, Collaboration, Central repository. 
I mean, what, you expect me to highlight a sentence and comment on it to suggest to another writer that she swaps out one word? That's a waste of time. editing and suggesting mode) that I had in Google Docs and were essential to my job as a writer/editor. It's definitely better as a document creator than a project management tool, but even then there are some essential features (i.e. But it also sucks at being a document creator. It lacks the collaborative project management features it needs to make it a successful project management tool. For example, I want to be able to assign cards in an Kanban board to other people, like you would in Asana or Trello. Quip is frustrating because it's really clunky and restrictive. CONSĪt my last job, I used a combination of Google Docs and Asana to manage projects and LOVED it. Kind of nice that you can create a document and a spreadsheet in one place. But really, these are all just small things that are kind of cool, but don't add to the overall usefulness of this software. Like, being able to insert a Kanban board inside a doc isn't something I've seen elsewhere. Google Docs) or project/work management software (i.e. Not a whole lot, but I suppose there are a couple of unique features, compared to document creators (i.e. And enough so that I'd totally risk rocking the boat to get them to transfer to something better. I have to use it because my team does, but I wish I didn't have to. It supports basic data representation objects like tables, pictures, worksheets but if you want audio etc, you want to goto powerpoint. If you are looking for a platform to create animated documents, quip might not be your friend. It behaves odd when creating tables and adding pictures (like indentation sometimes screws up). Onboarding and gettting used to the platform is really easy. You can create an excel sheet (or multiple sheets) in quip itself. You could easily import the content into a wiki or a word document with all the formatting intact. I have found Google Docs to be a bit slower than quip. It is so easy to mark your favorite folders and share it across teams. I could easily create documents that are public (i.e. Its easy to create public/private versions. I love quip as its the only good version controlled platform for taking notes that I have found in my industry experience of over 5 years. We use quip to track our updates, design documents etc and it has proven to be a really powerful platform for the same. 
And MS365 Personal plan’s monthly plan costs $6.99 only. It is rather expensive as it costs $70 per month for a single user. Microsoft Excel has 6 different plans: Microsoft 365 Business Basic (For Business) at 5.00. Explore pricing tiers and compare pricing against other Spreadsheets Software. The pricing for Microsoft Excel starts at 5.0 per user per month. Tableau is better used for creating graphics and trends. Check out the detailed pricing information for Microsoft Excel. TableauĮxcel is spreadsheet software used to perform complex calculations, data analysis, and more. 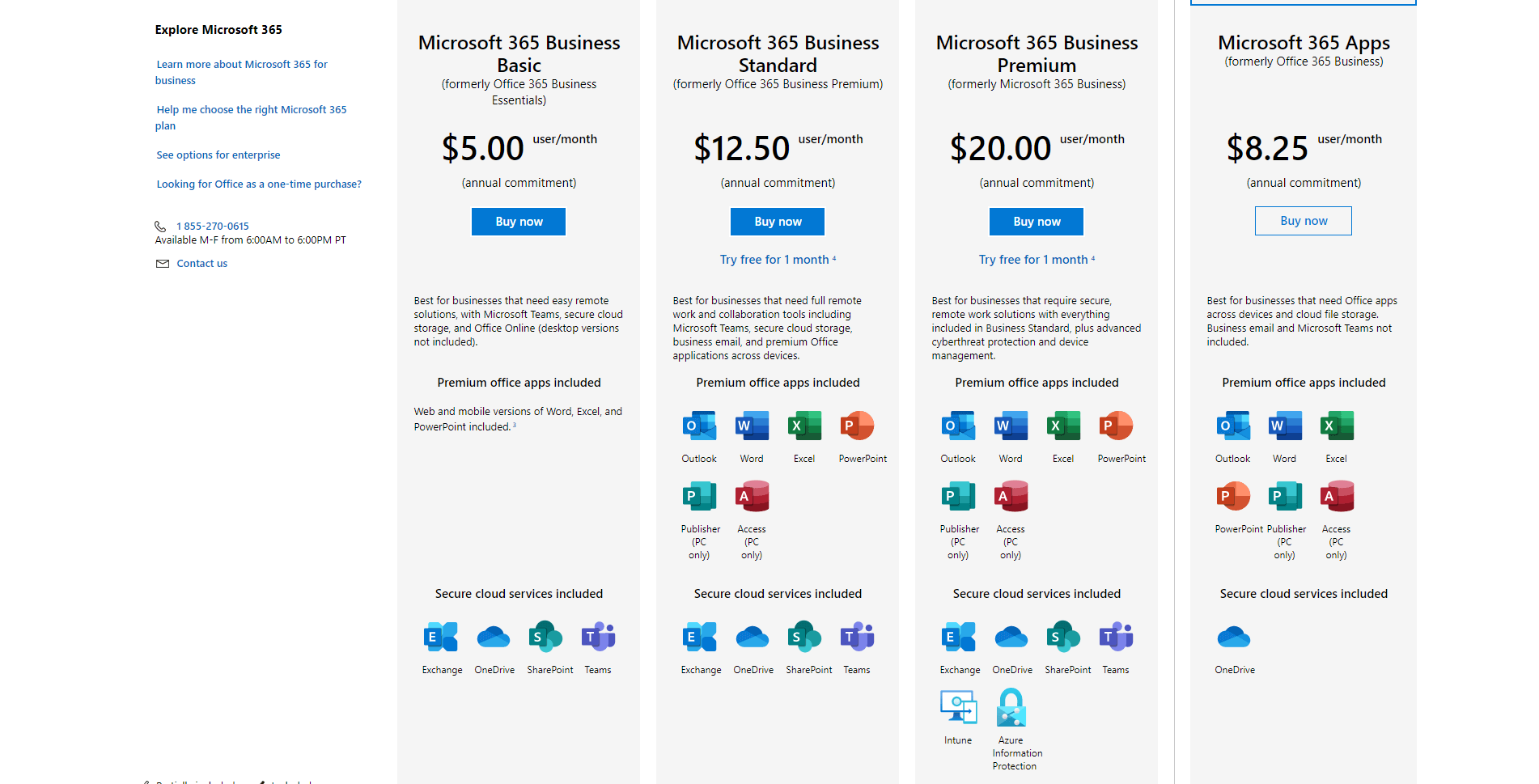
It offers four different pricing plans ranging from $0 to $15 per month.Įxcel is more famous for its pivot tables and power query and is priced at $6.99 per month. If your version of Excel doesnt have data types or the STOCKHISTORY function, dont worry, there is still an easy way to get the gold prices. A Microsoft 365 Personal subscription costs 69.99 per year and includes access to Microsoft Word, Excel, PowerPoint. The Smartsheet application is most commonly used for streamlining workflow and tracking tasks. Explore digital downloads for Microsoft Office 365 and MS Office Suite 2021 at Staples Canada, for business or personal use. Its Personal plan is free, while the other business plans range from $5-19 yearly □Įxcel comes as a part of the MS365 subscription service and is priced at $69.99 per year. ClickUpĬlickup software comes in a variety of pricing plans. Excel in MS365 needs to be subscribed to and costs $69.99/year. Google Sheets is a free online program commonly used for active collaboration. With Microsoft 365 you get Excel plus Word, PowerPoint, Outlook, and 1 TB of cloud storage per person for all your files, music, photos, videos, and more. Excel is better used to visualize data and perform data analysis. Google Sheets is a great software if you want to work on an Excel file with your team in real time. In contrast, MS365 Personal plan costs $69.99/year, but it is a powerful tool for calculations. But if you have to perform complex calculations and handle extensive data, Excel is your best bet.Īpple Numbers is free and is compatible with both Mac and PC. It offers a wide variety of templates to choose from and functions to use. 
Apple NumbersĪpple Numbers is great if you want a spreadsheet for simple calculations. Let’s see a quick comparison between the pricing plans of Excel and some other famous spreadsheet software □ Excel vs. 
You will also receive an email with your license purchase details (to the address indicated along with the information on your personal account). 
You will see a detailed guide on how to use the software. Once your payment is cleared, your device's IMEI or Serial Number will be automatically added to the service database, and you can use the software right away. Please check iRemove Software customer reviews on TrustPilot and rate our service to help us grow, develop, and self-improve.īy buying the license, you agree to the service operating conditions. iRemove Software is ranked #1 in the 'Cell Phone Recycling and Unlocking Service' category on Trustpilot. We value each client and are always happy to receive your feedback. Our service provides 100% refund guarantee. Please get in touch with our Support if you are willing to use another payment method. We accept several payment methods, including Visa, Mastercard, American Express, BTC\ETH\USDT cryptocurrency, Apple Pay, and Google Pay. Enter your email into the form and make payment. The software will automatically fill out the information about your locked device in the order form, namely its IMEI or Serial Number. If your device is supported by iRemove software, you can move to the payment page. Order iRemove iCloud Unlock Software Licence There is no need to download it as separate software. Checkra1n jailbreak is already built into the iRemove software. Checkra1n jailbreak is compatible with most iOS devices from iPhone 4S (A5 chip) and up to iPhone 8 |
 RSS Feed
RSS Feed
